 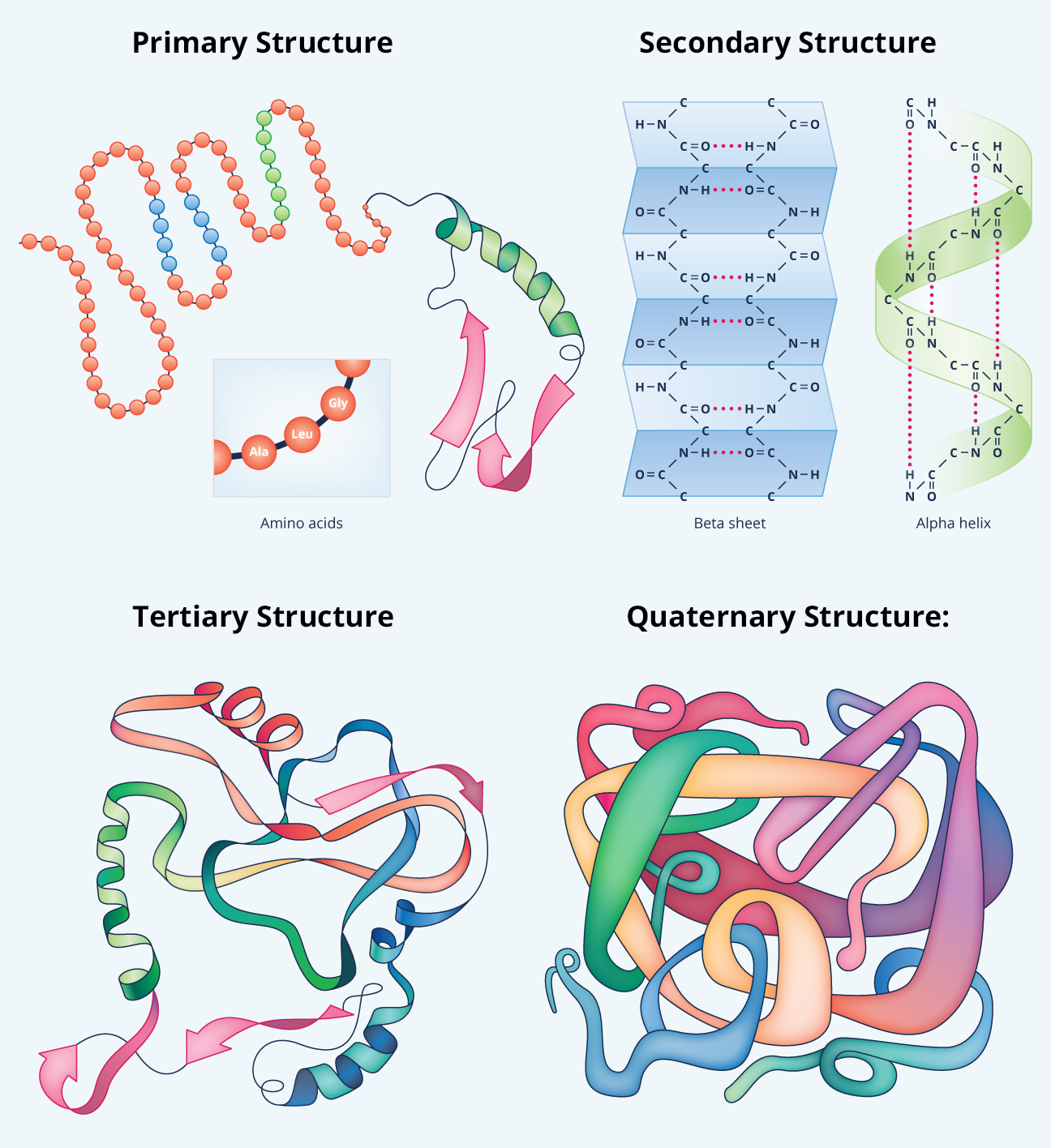
The sequence of amino acids in a protein is specified by the nucleotide sequence of the segment of DNA containing the gene that codes for that protein. Another important aspect of the primary structure is the sequence or order of amino acids in the polypeptide chain. The amino acids in a protein ✶ are linked together by a specific type of covalent bond, called a peptide bond, that exists between adjacent amino acids in the polypeptide chain. The primary structure is important to the protein's unique three-dimensional structure, its mechanism of action, and its relationship to other proteins with similar physiological roles. The primary structure refers to the amino acid sequence of a protein. The compression of amino acid sequences is a comparatively challenging task.There are four recognized levels of protein structure: primary, secondary, tertiary, and quaternary. Intermolecular transesterification can transfer a whole segment from one polypeptide to another, as is seen in the Hedgehog protein autoprocessing.In inteins, the new ester bond is broken by an intramolecular attack by the soon-to-be C-terminal asparagine. Intramolecular transesterification, resulting in a branched polypeptide.This pyruvoyl group may be used as a covalently attached catalytic cofactor in some enzymes, especially decarboxylases such as S-adenosylmethionine decarboxylase (SAMDC) that exploit the electron-withdrawing power of the pyruvoyl group. A β-elimination reaction also splits the chain, but results in a pyruvoyl group at the new N-terminus.This is seen in the maturation of glycosylasparaginase. 
Simple hydrolysis will split the polypeptide chain, where the displaced amino group becomes the new N-terminus.The ester/thioester bond can be resolved in several ways: This chemical reaction is called an N-O acyl shift. However, additional molecular interactions may render the amide form less stable the amino group is expelled instead, resulting in an ester (Ser/Thr) or thioester (Cys) bond in place of the peptide bond. This intermediate tends to revert to the amide form, expelling the attacking group, since the amide form is usually favored by free energy, (presumably due to the strong resonance stabilization of the peptide group). Typically, the hydroxyl group of a serine (rarely, threonine) or the thiol group of a cysteine residue will attack the carbonyl carbon of the preceding peptide bond, forming a tetrahedrally bonded intermediate. Some proteins even have the power to cleave themselves. The protein is activated by cleaving off the inhibitory peptide. Proteins are often synthesized in an inactive precursor form typically, an N-terminal or C-terminal segment blocks the active site of the protein, inhibiting its function. In addition to those listed above, the most important modification of primary structure is peptide cleavage (by chemical hydrolysis or by proteases). Many other chemical reactions (e.g., cyanylation) have been applied to proteins by chemists, although they are not found in biological systems. Most of the polypeptide modifications listed above occur post-translationally, i.e., after the protein has been synthesized on the ribosome, typically occurring in the endoplasmic reticulum, a subcellular organelle of the eukaryotic cell. Ubiquitin is the most common of these, and usually signals that the ubiquitin-tagged protein should be degraded. Various full-length, folded proteins can be attached at their C-termini to the sidechain ammonium groups of lysines of other proteins. This modification is a target for the powerful toxins of disparate bacteria, e.g., Vibrio cholerae, Corynebacterium diphtheriae and Bordetella pertussis. The large ADP-ribosyl group can be transferred to several types of side chains within proteins, with heterogeneous effects. This is used to strengthen the binding to "hard" metal ions such as calcium. Unlike the GPI and myritoyl anchors, these groups are not necessarily added at the termini.Ī relatively rare modification that adds an extra carboxylate group (and, hence, a double negative charge) to a glutamate side chain, producing a Gla residue. In particular, the L-amino acids normally found in proteins can spontaneously isomerize at the C α atom of cysteine residues to anchor proteins to cellular membranes. Although it does not change the sequence, it does affect the chemical properties of the sequence. 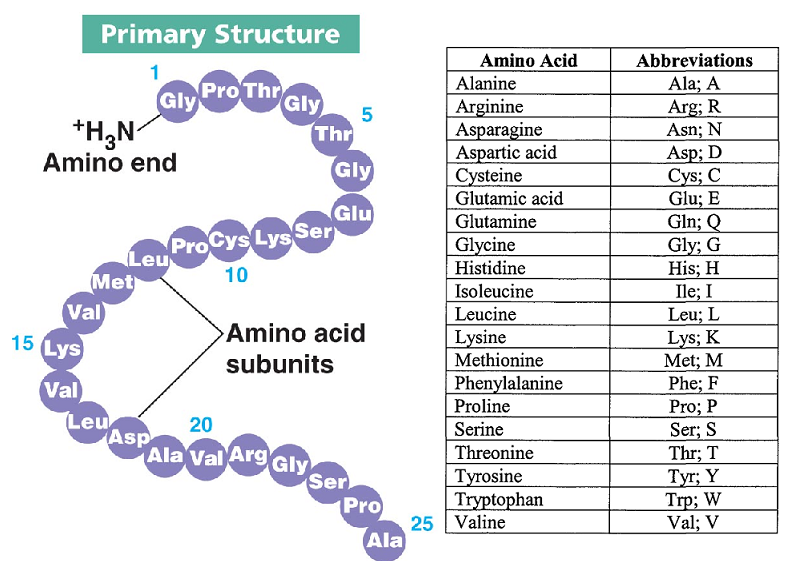
The chiral centers of a polypeptide chain can undergo racemization. However, proteins can become cross-linked, most commonly by disulfide bonds, and the primary structure also requires specifying the cross-linking atoms, e.g., specifying the cysteines involved in the protein's disulfide bonds. 
In general, polypeptides are unbranched polymers, so their primary structure can often be specified by the sequence of amino acids along their backbone. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
